Civil Lawsuits vs. VICP
When someone believes they were injured by a vaccine, it is common to think that filing a civil lawsuit—especially one decided by a jury—is the best way to seek justice and compensation. News stories about large pharmaceutical verdicts and settlements reinforce that belief.
In reality, vaccine injury claims operate very differently from most personal injury cases. Understanding the differences between civil lawsuits and the Vaccine Injury Compensation Program (VICP) helps explain why outcomes in vaccine cases rarely match public expectations.
Why Civil Litigation Feels Like the Better Option
Civil lawsuits feel familiar. People understand jury trials, courtroom storytelling, and the idea of holding a company legally responsible. High-profile pharmaceutical cases are widely reported and easy to remember.
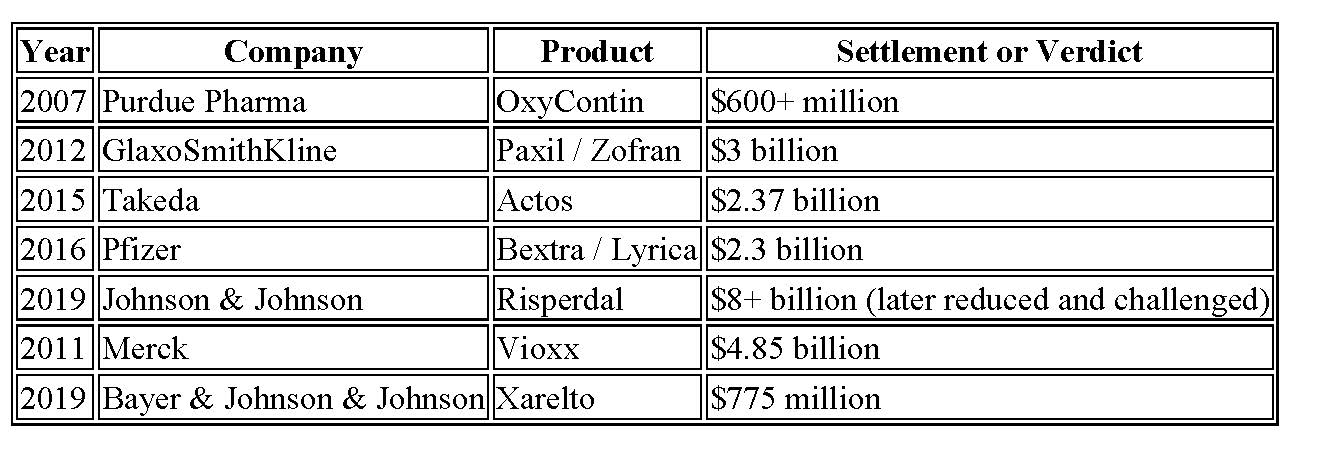
Examples often cited include:
These cases shape public perception—but they leave out critical context.
Each involved many years of litigation, often more than a decade, thousands of individual cases, extensive discovery, expert battles, appeals, and millions of dollars in legal costs advanced by plaintiffs’ attorneys. Pharmaceutical companies did not simply concede liability; they fought aggressively at every stage.
For every mass tort that ultimately results in a large settlement, many others fail—dismissed early, abandoned after expert testimony is excluded, or resolved for minimal amounts to end the litigation. Companies with deep pockets also have the resources to defend cases for years and win through delay and attrition.
This reality is even more pronounced in vaccine injury cases.
Burden of Proof
Civil Lawsuits
In a civil lawsuit, it is not enough to show that someone was injured after receiving a vaccine. The injured person must first prove that the vaccine itself was legally defective, and then prove that the defect caused the injury.
This is a very high standard.
Proving a Defect
To succeed in a civil vaccine injury case, a plaintiff must prove one of the following:
the vaccine was poorly designed,
the vaccine was manufactured incorrectly, or
the manufacturer failed to warn about known risks.
Each is difficult to establish in the vaccine context.
Vaccines are among the most closely studied and regulated medical products in the world. Hundreds of millions of vaccines are administered each year in the United States, and billions worldwide. The overwhelming majority of people receive vaccines without serious injury.
Because vaccines are used so widely and safely, courts often conclude that the design of the vaccine is not the problem when a rare injury occurs in an individual. The sheer volume of successful vaccinations strongly suggests that these injuries are uncommon biological responses rather than evidence of a flawed product.
Known risks are typically disclosed in the FDA-approved vaccine package insert. For that reason, failure-to-warn claims often fail early, because the risk was already disclosed. Design-defect claims face similar obstacles, as courts recognize that all medical products involve some unavoidable risks that must be balanced against significant public-health benefits.
Manufacturing-defect claims are possible, but they require proof that a specific dose deviated from approved standards—evidence that is rarely available.
If a defect cannot be proven, the civil case ends, regardless of the severity of the injury.
Proving Causation in Civil Court
Even if a defect could be shown, civil courts usually require large scientific studies demonstrating that the vaccine increases the risk of the specific injury alleged.
This creates another major barrier.
Vaccine injuries are rare. Because they occur infrequently, large population-based studies often do not exist. In many situations, it would be impossible—or unethical—to conduct the types of experiments courts expect, such as studies that intentionally expose people to potential harm.
When courts determine that the scientific evidence is insufficient, they often exclude expert testimony. When that happens, cases are dismissed before a jury ever hears the evidence.
Why Civil Lawsuits Usually Fail for Vaccine Injuries
Civil lawsuits fail in vaccine injury cases not because injuries never happen, but because the legal system requires proof that medical science often cannot provide.
Many vaccine injuries involve the immune system. Explaining how a vaccine can trigger an autoimmune or neurological injury often requires discussion of advanced immunology, including how immune responses become dysregulated or mistakenly target the body’s own tissues. These mechanisms are still being studied and cannot be ethically tested in humans.
Doctors may be able to explain why a vaccine can cause an injury and why it did cause an injury in a specific patient. Civil courts, however, often demand a level of scientific certainty that medicine itself cannot offer.
The Role of Daubert Standards
In civil cases, expert testimony must meet Daubert standards, which require judges to exclude expert opinions unless they are based on reliable methods, have been adequately tested, and are generally accepted in the scientific community.
Because many vaccine injuries are rare and individualized, there are often no large, controlled studies proving that a particular vaccine causes a particular injury in every case. Courts may therefore view expert opinions based on clinical judgment, biological plausibility, and patient-specific evidence as too speculative.
When expert testimony is excluded under Daubert, the case typically ends immediately, often without a jury ever hearing the claim.
Why Congress Created a Different System
Congress recognized this problem decades ago. Lawmakers understood that forcing injured individuals to prove product defects and large-scale scientific causation in civil court would leave most vaccine-injured people with no realistic path to compensation, even when injuries were real and life-altering.
The Vaccine Injury Compensation Program was created to address this mismatch between traditional tort law and scientific reality.
The Vaccine Injury Compensation Program (VICP)
The VICP operates under a no-fault framework. Petitioners do not need to prove negligence, misconduct, or that a vaccine was defective.
Instead, the focus is on whether:
a covered vaccine was administered, and
the individual suffered a vaccine-related injury.
Table Injuries: Presumed Causation
Some injuries are listed on the Vaccine Injury Table. These injuries are included because medical research generally supports a causal link between the vaccine and the injury when it occurs within a specific timeframe.
When a petitioner shows that:
a covered vaccine was given, and
a listed injury occurred within the required timeframe,
the law presumes that the vaccine caused the injury.
This presumption exists to streamline claims where the science already supports a connection. It does not mean the injury is common; it means the relationship is well understood.
Causation-in-Fact Claims: Most VICP Cases
Most VICP cases are causation-in-fact claims, meaning the injury is not listed on the Vaccine Injury Table or did not occur within the required timeframe.
These claims involve serious conditions such as:
Acute Disseminated Encephalomyelitis (ADEM)
Transverse Myelitis (TM)
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
Optic Neuritis
Neuromyelitis Optica (NMO)
Immune Thrombocytopenic Purpura (ITP)
Multiple Sclerosis (MS)
Bell’s Palsy
Vasculitis
In these cases, petitioners must show—using medical records and expert opinions—that the vaccine more likely than not caused the injury. Importantly, they do not need to prove a defective product or rely on large population studies.
This standard exists because Congress recognized that rare injuries would never be compensated if those requirements were imposed.
Cost, Risk, and Predictability
Civil lawsuits are expensive, risky, and often unpredictable. People can spend years in court and still recover nothing.
Under the VICP, reasonable attorneys’ fees and costs may be awarded even if a claim does not succeed, as long as statutory requirements are met. This greatly reduces financial risk and allows injured individuals to pursue claims they could not afford in civil court. This allows lawyers to represent injured victims no matter how big or small the injury is. Legal representation can cast a wide net to all those injured by vaccines to ensure anyone with a viable claim has her case reviewed, evaluated and if viable, fully prosecuted.
Petitioner Compensation
Civil lawsuits may promise large verdicts, but most vaccine cases never reach a damages phase.
The VICP provides compensation for:
medical expenses,
lost wages,
pain and suffering (up to $250,000), and
death benefits.
Compensation is paid from a dedicated trust fund—not by vaccine manufacturers.
What This Means for Vaccine Injured Individuals
Civil lawsuits and the VICP are not two versions of the same system. They are designed to address very different problems.
For vaccine injuries, the VICP was created to match how vaccines are used, how injuries occur, and what science can realistically prove.
Understanding this distinction allows injured individuals, policymakers, and the public to engage in informed discussions based on reality rather than headlines.
Final Perspective
Civil lawsuits and the VICP are not about choosing sides. They are about choosing the system that fits the problem.
Clear understanding—not rhetoric—is what allows honest conversation about vaccine injury compensation and public health.
Advocacy for the civil tort system as the “best” venue for vaccine injury claims often reflects a misunderstanding of how vaccine cases actually function in court—or an emphasis on incentives that do not align with the realities faced by injured individuals. Civil litigation can be attractive because it promises jury trials, expansive discovery, public filings, and the possibility of large verdicts. It also offers opportunities to generate headlines, pursue broad discovery into corporate records, advance policy arguments through litigation, or sustain prolonged legal battles that are largely disconnected from whether an injured person will ultimately be compensated.
Those features may serve strategic, political, or professional objectives. They rarely serve the practical interests of individuals with rare, medically complex vaccine injuries who need a realistic path to compensation.
The Vaccine Injury Compensation Program was created precisely because traditional tort litigation proved ill-suited to address these cases. It reflects a policy judgment that vaccine injuries require a system grounded in medical evidence, scientific uncertainty, and fairness—rather than fault-finding and attrition.
Understanding that distinction is essential to meaningful discussion of vaccine injury claims, informed policy reform, and the preservation of both public trust and public health.